Articles:
Notes:
Component of strawberry aroma, guava fruit and peel (Psidium guajava), pineapple, yellow passion fruit and other fruits. Also present in white wine and mussels. Flavouring ingredient
| CAS Number: | 10544-63-5 | 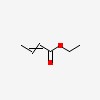 3D/inchi 3D/inchi
|
| ECHA EINECS - REACH Pre-Reg: | 234-125-9 | |
| FDA UNII: | Search | |
| Beilstein Number: | 1720418 | |
| CoE Number: | 2244 | |
| XlogP3-AA: | 1.30 (est) | |
| Molecular Weight: | 114.14410000 | |
| Formula: | C6 H10 O2 | |
| NMR Predictor: | Predict (works with chrome, Edge or firefox) | |
| Also(can) Contains: | ethyl (E)-2-crotonate | |
Category: flavor and fragrance agents
US / EU / FDA / JECFA / FEMA / FLAVIS / Scholar / Patent Information:
| Google Scholar: | Search |
| Google Books: | Search |
| Google Scholar: with word "volatile" | Search |
| Google Scholar: with word "flavor" | Search |
| Google Scholar: with word "odor" | Search |
| Perfumer and Flavorist: | Search |
| Google Patents: | Search |
| US Patents: | Search |
| EU Patents: | Search |
| Pubchem Patents: | Search |
| PubMed: | Search |
| NCBI: | Search |
| JECFA Food Flavoring: | 1806 ethyl trans-2-butenoate |
| FEMA Number: | 3486 ethyl trans-2-butenoate |
| FDA: | No longer provide for the use of these seven synthetic flavoring substances |
| FDA Mainterm (SATF): | 623-70-1 ; ETHYL CROTONATE |
| FDA Regulation: | |
| FDA PART 172 -- FOOD ADDITIVES PERMITTED FOR DIRECT ADDITION TO FOOD FOR HUMAN CONSUMPTION Subpart F--Flavoring Agents and Related Substances Sec. 172.515 Synthetic flavoring substances and adjuvants. | |
Physical Properties:
| Appearance: | colorless clear liquid (est) |
| Assay: | 98.00 to 100.00 % |
| Food Chemicals Codex Listed: | Yes |
| Specific Gravity: | 0.91400 to 0.92000 @ 20.00 °C. |
| Pounds per Gallon - (est).: | 7.614 to 7.664 |
| Refractive Index: | 1.42300 to 1.42800 @ 20.00 °C. |
| Boiling Point: | 136.00 to 137.00 °C. @ 760.00 mm Hg |
| Acid Value: | 2.00 max. KOH/g |
| Vapor Pressure: | 6.866000 mmHg @ 25.00 °C. (est) |
| Flash Point: | 36.00 °F. TCC ( 2.22 °C. ) |
| logP (o/w): | 1.806 (est) |
| Soluble in: | |
| alcohol | |
| water, slightly | |
| water, 4259 mg/L @ 25 °C (est) | |
| Insoluble in: | |
| water | |
| Similar Items: note | |
| allyl crotonate | |
| isoamyl crotonate | |
| apple crotonate | |
| benzyl crotonate | |
| butyl (E)-2-crotonate | |
| citronellyl crotonate | |
| cyclohexyl crotonate | |
| dimethyl benzyl carbinyl crotonate | |
| ethyl (E)-2-crotonate | |
| fig crotonate | |
| geranyl crotonate | |
| heptyl crotonate | |
| (Z)-3-hexen-1-yl (E)-crotonate | |
| (E)-3-hexen-1-yl crotonate | |
| hexyl (E)-crotonate | |
| octyl crotonate | |
| phenethyl crotonate | |
| 3-phenyl propyl crotonate | |
| plum crotonate | |
| isopropyl (E)-crotonate | |
| propyl crotonate | |
| isopropyl 3-methyl thiocrotonate | |
Organoleptic Properties:
| Odor Type: fermented | |
| pungent fruity acidic estery | |
| Odor Description: at 10.00 % in dipropylene glycol. | pungent fruity acrid ester |
| pungent sharp rummy cognac pineapple fruity meaty | |
| Odor Description: | Pungent, sharp, rum- and cognac-Iike, wth tinny, pineapple, fruity and meaty nuances Mosciano, Gerard P&F 20, No. 6, 49, (1995) |
| Flavor Type: rummy | |
| rummy cognac pungent caramellic fruity | |
| Taste Description: at 10.00 ppm. | Rum, cognac and pungent with caramellic and fruity nuances Mosciano, Gerard P&F 20, No. 6, 49, (1995) |
| Odor and/or flavor descriptions from others (if found). | |
Cosmetic Information:
| CosIng: | cosmetic data |
| Cosmetic Uses: |
perfuming agents |
Suppliers:
| Apple Flavor & Fragrance |
| Ethyl crotonate |
| BOC Sciences |
| For experimental / research use only. |
| Ethyl crotonate 95% |
| CJ Latta & Associates |
| ETHYL CROTONATE |
| Diffusions Aromatiques |
| ETHYL CROTONATE |
| Ernesto Ventós |
| ETHYL CROTONATE |
| Godavari Biorefineries |
| Ethyl Crotonate |
| Grau Aromatics |
| ETHYL-CROTONATE FCC
NI, Kosher |
| Indukern F&F |
| ETHYL CROTONATE
Odor: ETHEREAL, POWERFUL, FRUITY |
| Inoue Perfumery |
| ETHYL CROTONATE |
| Kingchem Laboratories |
| ETHYL CROTONATE (T2 BUTENOATE)
Odor: Sweet, ethereal |
| Lluch Essence |
| ETHYL CROTONATE
Odor: FRUITY, GASSY |
| SRS Aromatics |
| ETHYL CROTONATE FCC |
| WholeChem |
| Ethyl crotonate |
Safety Information:
| European information : | |
| Most important hazard(s): | |
| Xi - Irritant | |
|
R 11 - Highly flammable. R 36/37/38 - Irritating to eyes, respiratory system, and skin. S 02 - Keep out of the reach of children. S 16 - Keep away from sources of ignition - No Smoking. S 23 - Do not breath vapour. S 24/25 - Avoid contact with skin and eyes. S 26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S 36/37/39 - Wear suitable clothing, gloves and eye/face protection. | |
| Hazards identification | |
| Classification of the substance or mixture | |
| GHS Classification in accordance with 29 CFR 1910 (OSHA HCS) | |
| None found. | |
| GHS Label elements, including precautionary statements | |
| Pictogram | |
| Hazard statement(s) | |
| None found. | |
| Precautionary statement(s) | |
| None found. | |
| Oral/Parenteral Toxicity: | |
|
oral-rat LD50 3000 mg/kg Nippon Eiseigaku Zasshi. Japanese Journal of Hygiene. Vol. 34, Pg. 183, 1979. | |
| Dermal Toxicity: | |
|
skin-guinea pig LD50 > 10 mL/kg Journal of Industrial Hygiene and Toxicology. Vol. 26, Pg. 269, 1944. | |
| Inhalation Toxicity: | |
| Not determined | |
Safety in Use Information:
| Category: | flavor and fragrance agents | ||
| RIFM Fragrance Material Safety Assessment: Search | |||
| IFRA Code of Practice Notification of the 49th Amendment to the IFRA Code of Practice | |||
| Recommendation for ethyl crotonate usage levels up to: | |||
| 6.0000 % in the fragrance concentrate. | |||
| Use levels for FEMA GRAS flavoring substances on which the FEMA Expert Panel based its judgments that the substances are generally recognized as safe (GRAS). | |||
| The Expert Panel also publishes separate extensive reviews of scientific information on all FEMA GRAS flavoring substances and can be found at FEMA Flavor Ingredient Library | |||
| publication number: 10 | |||
| Click here to view publication 10 | |||
| average usual ppm | average maximum ppm | ||
| baked goods: | - | 20.70000 | |
| beverages(nonalcoholic): | - | 3.00000 | |
| beverages(alcoholic): | - | 4.00000 | |
| breakfast cereal: | - | - | |
| cheese: | - | - | |
| chewing gum: | - | - | |
| condiments / relishes: | - | - | |
| confectionery froastings: | - | 16.10000 | |
| egg products: | - | - | |
| fats / oils: | - | - | |
| fish products: | - | - | |
| frozen dairy: | - | 8.40000 | |
| fruit ices: | - | 8.40000 | |
| gelatins / puddings: | - | 5.71000 | |
| granulated sugar: | - | - | |
| gravies: | - | - | |
| hard candy: | - | - | |
| imitation dairy: | - | - | |
| instant coffee / tea: | - | - | |
| jams / jellies: | - | - | |
| meat products: | - | - | |
| milk products: | - | - | |
| nut products: | - | - | |
| other grains: | - | - | |
| poultry: | - | - | |
| processed fruits: | - | - | |
| processed vegetables: | - | - | |
| reconstituted vegetables: | - | - | |
| seasonings / flavors: | - | - | |
| snack foods: | - | - | |
| soft candy: | - | - | |
| soups: | - | - | |
| sugar substitutes: | - | - | |
| sweet sauces: | - | - | |
Safety References:
| Flavor & Extract Manufacturers Association (FEMA) reference(s): | |
| The FEMA GRAS assessment of alpha,beta-unsaturated aldehydes and related substances used as flavor ingredients. View pdf | |
| EPI System: | View |
| AIDS Citations: | Search |
| Cancer Citations: | Search |
| Toxicology Citations: | Search |
| EPA Substance Registry Services (TSCA): | 10544-63-5 |
| EPA ACToR: | Toxicology Data |
| EPA Substance Registry Services (SRS): | Registry |
| Laboratory Chemical Safety Summary : | 12191 |
| National Institute of Allergy and Infectious Diseases: | Data |
| WGK Germany: | 2 |
| ethyl but-2-enoate | |
| Chemidplus: | 0010544635 |
| EPA/NOAA CAMEO: | hazardous materials |
References:
| ethyl but-2-enoate | |
| NIST Chemistry WebBook: | Search Inchi |
| Canada Domestic Sub. List: | 10544-63-5 |
| Pubchem (cid): | 12191 |
| Pubchem (sid): | 135018036 |
| Pherobase: | View |
Other Information:
| (IUPAC): | Atomic Weights of the Elements 2011 (pdf) |
| Videos: | The Periodic Table of Videos |
| tgsc: | Atomic Weights use for this web site |
| (IUPAC): | Periodic Table of the Elements |
| FDA Substances Added to Food (formerly EAFUS): | View |
| HMDB (The Human Metabolome Database): | HMDB39581 |
| FooDB: | FDB019204 |
| YMDB (Yeast Metabolome Database): | YMDB01379 |
| Export Tariff Code: | 2916.19.7000 |
| VCF-Online: | VCF Volatile Compounds in Food |
| ChemSpider: | View |
Potential Blenders and core components note
Potential Uses:
| blueberry | FR | |
| cherry | FR | |
| citrus | FR | |
| cucumber | FR | |
| guava | FR | |
| orange | FR | |
| raspberry | FR | |
| strawberry | FR |
Occurrence (nature, food, other): note
| guava fruit Search Trop Picture | |
| michelia figo Search Trop Picture | |
| passion fruit yellow Search Trop Picture | |
| strawberry wild strawberry fruit Search Trop Picture | |
| wine white wine Search Picture |
Synonyms:
| 2- | butenoic acid ethyl ester |
| 2- | butenoic acid, ethyl ester |
| crotonic acid ethyl ester | |
| ethyl 2-butenoate | |
| ethyl but-2-enoate | |
| ethyl crotonate (T2 butenoate) | |
| ethyl-2-butenoate | |
| ethyl-crotonate FCC |






