Articles:
2-propenal, 3-(4-methoxyphenyl)-
Notes:
Isol. from oil of tarragon (Artemisia dracunculus) and other oils. Flavouring ingredient
| CAS Number: | 1963-36-6 | 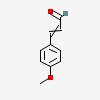 3D/inchi 3D/inchi
|
| Other(deleted CASRN): | 1619932-83-0 | |
| ECHA EINECS - REACH Pre-Reg: | 217-807-0 | |
| FDA UNII: | Search | |
| MDL: | MFCD00017343 | |
| CoE Number: | 11919 | |
| XlogP3: | 1.80 (est) | |
| Molecular Weight: | 162.18810000 | |
| Formula: | C10 H10 O2 | |
| BioActivity Summary: | listing | |
| NMR Predictor: | Predict (works with chrome, Edge or firefox) | |
Category: flavor and fragrance agents
US / EU / FDA / JECFA / FEMA / FLAVIS / Scholar / Patent Information:
| Google Scholar: | Search |
| Google Books: | Search |
| Google Scholar: with word "volatile" | Search |
| Google Scholar: with word "flavor" | Search |
| Google Scholar: with word "odor" | Search |
| Perfumer and Flavorist: | Search |
| Google Patents: | Search |
| US Patents: | Search |
| EU Patents: | Search |
| Pubchem Patents: | Search |
| PubMed: | Search |
| NCBI: | Search |
| JECFA Food Flavoring: | 687 p-methoxycinnamaldehyde |
| DG SANTE Food Flavourings: | 05.118 4-methoxycinnamaldehyde |
| FEMA Number: | 3567 p-methoxycinnamaldehyde |
| FDA: | No longer provide for the use of these seven synthetic flavoring substances |
| FDA Mainterm (SATF): | 1963-36-6 ; P-METHOXYCINNAMALDEHYDE |
Physical Properties:
| Appearance: | yellow crystals (est) |
| Assay: | 96.00 to 100.00 % |
| Food Chemicals Codex Listed: | No |
| Melting Point: | 44.00 to 45.00 °C. @ 760.00 mm Hg |
| Boiling Point: | 160.00 °C. @ 3.00 mm Hg |
| Boiling Point: | 308.00 to 309.00 °C. @ 760.00 mm Hg |
| Vapor Pressure: | 0.001000 mmHg @ 25.00 °C. (est) |
| Vapor Density: | >1 ( Air = 1 ) |
| Flash Point: | 230.00 °F. TCC ( 110.00 °C. ) |
| logP (o/w): | 1.953 (est) |
| Soluble in: | |
| alcohol | |
| water, 1598 mg/L @ 25 °C (est) | |
| Insoluble in: | |
| water | |
Organoleptic Properties:
| Odor Type: spicy | |
| spicy cinnamon sweet cherry vanilla floral | |
| Odor Description: at 10.00 % in dipropylene glycol. | spicy cinnamon sweet cherry vanilla floral |
| spicy cinnamon sweet cherry candy vanilla smoky | |
| Odor Description: at 0.50 %. | Spicy, cinnamon, sweet with a rich pleasant cherry candy note, vanilla and slightly smoky Mosciano, Gerard P&F 26, No. 2, 40, (2001) |
| Flavor Type: cherry | |
| cherry fleshy sweet cinnamyl aromatic | |
| Taste Description: at 3.00 - 20.00 ppm. | Cherry flesh, sweet, with a cinnamic dry out, slightly aromatic Mosciano, Gerard P&F 26, No. 2, 40, (2001) |
| Odor and/or flavor descriptions from others (if found). | |
| Bedoukian Research | |
| para-METHOXY CINNAMIC ALDEHYDE (PMCA) ≥98.0%, Kosher | |
| Odor Description: | A pleasant cinnamon note that is not overpowering Widely used in spice fragrances. |
| Taste Description: | spicy Adds a sweet, cinnamon note to many applications such as spice blends and oral hygiene flavors. |
| Alfrebro | |
| p-METHOXY-CINNAMALDEHYDE NATURAL | |
| Odor Description: | Warm, Cinnamon, Anise, Spicy |
Cosmetic Information:
| CosIng: | cosmetic data |
| Cosmetic Uses: |
perfuming agents |
Suppliers:
| Alfrebro |
| p-METHOXY-CINNAMALDEHYDE NATURAL
Odor: Warm, Cinnamon, Anise, Spicy |
| BOC Sciences |
| For experimental / research use only. |
| para-Methoxycinnamaldehyde 98.0% |
| M&U International |
| para-Methoxy Cinnamic Aldehyde |
| Penta International |
| P-METHOXY CINNAMIC ALDEHYDE NATURAL |
| Penta International |
| P-METHOXY CINNAMIC ALDEHYDE |
| TCI AMERICA |
| For experimental / research use only. |
| 4-Methoxycinnamaldehyde >97.0%(GC) |
Safety Information:
| European information : | |
| Most important hazard(s): | |
| Xi - Irritant | |
|
R 36/38 - Irritating to skin and eyes. S 02 - Keep out of the reach of children. S 24/25 - Avoid contact with skin and eyes. S 26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S 36 - Wear suitable protective clothing. | |
| Hazards identification | |
| Classification of the substance or mixture | |
| GHS Classification in accordance with 29 CFR 1910 (OSHA HCS) | |
| None found. | |
| GHS Label elements, including precautionary statements | |
| Pictogram | |
| Hazard statement(s) | |
| None found. | |
| Precautionary statement(s) | |
| None found. | |
| Oral/Parenteral Toxicity: | |
| Not determined | |
| Dermal Toxicity: | |
| Not determined | |
| Inhalation Toxicity: | |
| Not determined | |
Safety in Use Information:
| Category: | flavor and fragrance agents | ||
| RIFM Fragrance Material Safety Assessment: Search | |||
| IFRA Code of Practice Notification of the 49th Amendment to the IFRA Code of Practice | |||
| Recommendation for para-methoxycinnamaldehyde usage levels up to: | |||
| 3.0000 % in the fragrance concentrate. | |||
| Maximised Survey-derived Daily Intakes (MSDI-EU): | 0.037 (μg/capita/day) | ||
| Maximised Survey-derived Daily Intakes (MSDI-USA): | 0.01 (μg/capita/day) | ||
| Structure Class: | I | ||
| Use levels for FEMA GRAS flavoring substances on which the FEMA Expert Panel based its judgments that the substances are generally recognized as safe (GRAS). | |||
| The Expert Panel also publishes separate extensive reviews of scientific information on all FEMA GRAS flavoring substances and can be found at FEMA Flavor Ingredient Library | |||
| publication number: 11 | |||
| Click here to view publication 11 | |||
| average usual ppm | average maximum ppm | ||
| baked goods: | - | 5.00000 | |
| beverages(nonalcoholic): | - | - | |
| beverages(alcoholic): | - | - | |
| breakfast cereal: | - | - | |
| cheese: | - | - | |
| chewing gum: | - | - | |
| condiments / relishes: | - | 4.00000 | |
| confectionery froastings: | - | 5.00000 | |
| egg products: | - | - | |
| fats / oils: | - | - | |
| fish products: | - | - | |
| frozen dairy: | - | - | |
| fruit ices: | - | - | |
| gelatins / puddings: | - | 5.00000 | |
| granulated sugar: | - | - | |
| gravies: | - | - | |
| hard candy: | - | - | |
| imitation dairy: | - | - | |
| instant coffee / tea: | - | - | |
| jams / jellies: | - | 5.00000 | |
| meat products: | - | 2.00000 | |
| milk products: | - | 2.00000 | |
| nut products: | - | - | |
| other grains: | - | - | |
| poultry: | - | - | |
| processed fruits: | - | - | |
| processed vegetables: | - | - | |
| reconstituted vegetables: | - | - | |
| seasonings / flavors: | - | 5.00000 | |
| snack foods: | - | - | |
| soft candy: | - | - | |
| soups: | - | - | |
| sugar substitutes: | - | - | |
| sweet sauces: | - | 4.00000 | |
Safety References:
| Flavor & Extract Manufacturers Association (FEMA) reference(s): | |
| The FEMA GRAS assessment of cinnamyl derivatives used as flavor ingredients. View pdf | |
| European Food Safety Athority(EFSA): | Flavor usage levels; Subacute, Subchronic, Chronic and Carcinogenicity Studies; Developmental / Reproductive Toxicity Studies; Genotoxicity Studies... |
| European Food Safety Authority (EFSA) reference(s): | |
| Flavouring Group Evaluation 214: alpha,beta-Unsaturated aldehydes and precursors from chemical subgroup 3.1 of FGE.19: Cinnamyl derivatives[1] View page or View pdf | |
| Scientific Opinion on Flavouring Group Evaluation 68 (FGE.68): Consideration of cinnamyl alcohol and related flavouring agents evaluated by JECFA (55th meeting) evaluated by EFSA in FGE.15Rev1 (2008) View page or View pdf | |
| EPI System: | View |
| AIDS Citations: | Search |
| Cancer Citations: | Search |
| Toxicology Citations: | Search |
| EPA Substance Registry Services (TSCA): | 1963-36-6 |
| EPA ACToR: | Toxicology Data |
| EPA Substance Registry Services (SRS): | Registry |
| Laboratory Chemical Safety Summary : | 230995 |
| National Institute of Allergy and Infectious Diseases: | Data |
| WGK Germany: | 2 |
| 3-(4-methoxyphenyl)prop-2-enal | |
| Chemidplus: | 0001963366 |
References:
| 3-(4-methoxyphenyl)prop-2-enal | |
| NIST Chemistry WebBook: | Search Inchi |
| Canada Domestic Sub. List: | 1963-36-6 |
| Pubchem (cid): | 230995 |
| Pubchem (sid): | 135198987 |
| Pherobase: | View |
Other Information:
| (IUPAC): | Atomic Weights of the Elements 2011 (pdf) |
| Videos: | The Periodic Table of Videos |
| tgsc: | Atomic Weights use for this web site |
| (IUPAC): | Periodic Table of the Elements |
| FDA Substances Added to Food (formerly EAFUS): | View |
| Metabolomics Database: | Search |
| KEGG (GenomeNet): | C10475 |
| HMDB (The Human Metabolome Database): | HMDB32611 |
| FooDB: | FDB010552 |
| Export Tariff Code: | 2912.49.2600 |
| VCF-Online: | VCF Volatile Compounds in Food |
| ChemSpider: | View |
| Wikipedia: | View |
Potential Blenders and core components note
Potential Uses:
| cassia | FR | |
| cherry | FR | |
| cinnamon | FR | |
| floral | FR | |
| honey | FR | |
| licorice red licorice | FL | |
| maple | FR | |
| spice | FR | |
| tobacco | FR | |
| vanilla | FR |
Occurrence (nature, food, other): note
| anise star anise Search Trop Picture | |
| anise star anise plant Search Trop Picture | |
| basil flower oil Search Trop Picture | |
| potato baked potato Search Trop Picture | |
| potato tuber Search Trop Picture | |
| tarragon leaf oil Search Trop Picture | |
| tarragon oil Search Trop Picture | |
| tarragon seed Search Trop Picture | |
| tarragon seed oil Search Trop Picture | |
| tarragon shoot Search Trop Picture | |
| tarragon shoot oil Search Trop Picture |
Synonyms:
| p- | methoxy cinnamic aldehyde (PMCA) |
| p- | methoxy cinnamic aldehyde natural |
| p- | methoxy-cinnamaldehyde |
| 4- | methoxycinnamaldehyde |
| p- | methoxycinnamaldehyde |
| p- | methoxycinnamic aldehyde |
| para- | methoxycinnamic aldehyde |
| 3-(4- | methoxyphenyl)-2-propenal |
| 3-(4- | methoxyphenyl)acrylaldehyde |
| 3-(4- | methoxyphenyl)prop-2-enal |
| p- | methxoy cinnamic aldehyde |
| 2- | propenal, 3-(4-methoxyphenyl)- |




