Articles:
3-L-menthoxypropane-1,2-diol
Notes:
Physiol. cooling agent used in food and beverages
| CAS Number: | 87061-04-9 | 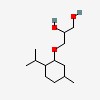 3D/inchi 3D/inchi
|
| Other(deleted CASRN): | 163271-16-7 | |
| ECHA EINECS - REACH Pre-Reg: | 289-296-2 | |
| FDA UNII: | Search | |
| Nikkaji Web: | J60.959E | |
| XlogP3: | 2.30 (est) | |
| Molecular Weight: | 230.34782000 | |
| Formula: | C13 H26 O3 | |
| NMR Predictor: | Predict (works with chrome, Edge or firefox) | |
| EFSA/JECFA Comments: | JECFA name: 3-L-Menthoxypropane-1,2-diol. Racemate. | |
Category: flavor and fragrance agents
US / EU / FDA / JECFA / FEMA / FLAVIS / Scholar / Patent Information:
| Google Scholar: | Search |
| Google Books: | Search |
| Google Scholar: with word "volatile" | Search |
| Google Scholar: with word "flavor" | Search |
| Google Scholar: with word "odor" | Search |
| Perfumer and Flavorist: | Search |
| Google Patents: | Search |
| US Patents: | Search |
| EU Patents: | Search |
| Pubchem Patents: | Search |
| PubMed: | Search |
| NCBI: | Search |
| JECFA Food Flavoring: | 1408 3-L-menthoxypropane-1,2-diol |
| DG SANTE Food Flavourings: | 02.224 3-(L-menthoxy)propane-1,2-diol |
| FEMA Number: | 3784 3-L-menthoxypropane-1,2-diol |
| FDA: | No longer provide for the use of these seven synthetic flavoring substances |
| FDA Mainterm (SATF): | 87061-04-9 ; 3-L-MENTHOXYPROPANE-1,2-DIOL |
Physical Properties:
| Appearance: | colorless clear viscous liquid (est) |
| Assay: | 99.00 to 100.00 % |
| Food Chemicals Codex Listed: | No |
| Specific Gravity: | 0.99000 to 1.00200 @ 25.00 °C. |
| Pounds per Gallon - (est).: | 8.238 to 8.338 |
| Refractive Index: | 1.47200 to 1.47600 @ 20.00 °C. |
| Optical Rotation: | -86.0 to -80.0 |
| Boiling Point: | 121.00 to 125.00 °C. @ 0.25 mm Hg |
| Boiling Point: | 361.00 to 362.00 °C. @ 760.00 mm Hg |
| Acid Value: | 1.00 max. KOH/g |
| Flash Point: | 343.00 °F. TCC ( 172.78 °C. ) |
| logP (o/w): | 2.625 (est) |
| Soluble in: | |
| alcohol | |
| water, very slightly | |
| water, 108.1 mg/L @ 25 °C (est) | |
| Insoluble in: | |
| water | |
Organoleptic Properties:
| Odor Type: minty | |
| Odor Strength: | low |
| Substantivity: | 81 hour(s) at 100.00 % |
| minty phenolic fruity jammy fruit dried fruit berry | |
| Odor Description: at 100.00 %. | very mild minty phenolic fruity jammy dried fruit berry Luebke, William tgsc, (2021) |
| Flavor Type: cooling | |
| cooling fresh aromatic mentholic eucalyptus | |
| Taste Description: | clean cooling fresh icy aromatic mentholic Luebke, William tgsc, (2021) |
| Odor and/or flavor descriptions from others (if found). | |
Cosmetic Information:
| CosIng: | cosmetic data |
| Cosmetic Uses: |
fragrance refreshing agents |
Suppliers:
| Apple Flavor & Fragrance |
| Cooling agent MOPD |
| BeYonde |
| Coolact Agent 10 (MOPD) |
| BOC Sciences |
| For experimental / research use only. |
| 3-[[5-Methyl-2-(1-methylethyl)cyclohexyl]oxy]propane-1,2-diol |
| Jinan Enlighten Chemical Technology(Wutong Aroma ) |
| 3-L-Menthoxypropane-1,2-diol, Kosherk |
| Lipo Chemicals |
| Coolact® 10
Odor: characteristic Use: Coolact® 10 is a sensory agent that provides a cooling effect to the skin. The product functions by directly stimulating the receptors at the nerve endings of the skin to produce a cooling sensation. Coolact® 10 provides a long lasting cooling effect. |
| O'Laughlin Industries |
| ARCTIC SW-10, TK-10 ? |
| OQEMA |
| Lansense 10 |
| Parchem |
| menthoxypropane diol |
| Penta International |
| 3-1-MENTHOXYPROPANE-1,2-DIOL |
| Shanghai Vigen Fine Chemical |
| 3-l-(p-menthan-3-yloxy)-1,2-propanediol
(Cooling Agent 10 ) |
| Takasago |
| Coolact 10 |
| The Fragrance Museum |
Safety Information:
| Hazards identification | |
| Classification of the substance or mixture | |
| GHS Classification in accordance with 29 CFR 1910 (OSHA HCS) | |
| None found. | |
| GHS Label elements, including precautionary statements | |
| Pictogram | |
| Hazard statement(s) | |
| None found. | |
| Precautionary statement(s) | |
| None found. | |
| Oral/Parenteral Toxicity: | |
| Not determined | |
| Dermal Toxicity: | |
| Not determined | |
| Inhalation Toxicity: | |
| Not determined | |
Safety in Use Information:
| Category: | flavor and fragrance agents | ||
| RIFM Fragrance Material Safety Assessment: Search | |||
| IFRA Code of Practice Notification of the 49th Amendment to the IFRA Code of Practice | |||
| Recommendation for menthoxypropane diol usage levels up to: | |||
| 3.0000 % in the fragrance concentrate. | |||
| Maximised Survey-derived Daily Intakes (MSDI-EU): | 4.10 (μg/capita/day) | ||
| Maximised Survey-derived Daily Intakes (MSDI-USA): | 789.00 (μg/capita/day) | ||
| Structure Class: | I | ||
| Use levels for FEMA GRAS flavoring substances on which the FEMA Expert Panel based its judgments that the substances are generally recognized as safe (GRAS). | |||
| The Expert Panel also publishes separate extensive reviews of scientific information on all FEMA GRAS flavoring substances and can be found at FEMA Flavor Ingredient Library | |||
| publication number: 16 | |||
| Click here to view publication 16 | |||
| average usual ppm | average maximum ppm | ||
| baked goods: | - | - | |
| beverages(nonalcoholic): | 300.00000 | 500.00000 | |
| beverages(alcoholic): | 300.00000 | 500.00000 | |
| breakfast cereal: | - | - | |
| cheese: | - | - | |
| chewing gum: | 3000.00000 | 4000.00000 | |
| condiments / relishes: | - | - | |
| confectionery froastings: | 100.00000 | 500.00000 | |
| egg products: | - | - | |
| fats / oils: | - | - | |
| fish products: | - | - | |
| frozen dairy: | - | - | |
| fruit ices: | - | - | |
| gelatins / puddings: | 500.00000 | 1000.00000 | |
| granulated sugar: | - | - | |
| gravies: | - | - | |
| hard candy: | 500.00000 | 500.00000 | |
| imitation dairy: | 100.00000 | 500.00000 | |
| instant coffee / tea: | - | - | |
| jams / jellies: | - | - | |
| meat products: | - | - | |
| milk products: | - | - | |
| nut products: | - | - | |
| other grains: | - | - | |
| poultry: | - | - | |
| processed fruits: | - | - | |
| processed vegetables: | - | - | |
| reconstituted vegetables: | - | - | |
| seasonings / flavors: | - | - | |
| snack foods: | - | - | |
| soft candy: | 100.00000 | 500.00000 | |
| soups: | - | - | |
| sugar substitutes: | - | - | |
| sweet sauces: | - | - | |
Safety References:
| Flavor & Extract Manufacturers Association (FEMA) reference(s): | |
| The FEMA GRAS assessment of alicyclic substances used as flavor ingredients. View pdf | |
| European Food Safety Athority(EFSA): | Flavor usage levels; Subacute, Subchronic, Chronic and Carcinogenicity Studies; Developmental / Reproductive Toxicity Studies; Genotoxicity Studies... |
| European Food Safety Authority (EFSA) reference(s): | |
| Flavouring Group Evaluation 23, Revision 1 (FGE.23Rev1): Aliphatic, alicyclic and aromatic ethers including anisole derivatives from chemical groups 15, 16, 26 and 30[1] - Opinion of the Scientific Panel on Food Additives, Flavourings, Processing Aids and Materials in Contact with Food View page or View pdf | |
| Flavouring Group Evaluation 9, Revision 1: (FGE.09 Rev1)[1] - Secondary alicyclic saturated and unsaturated alcohols, ketones and esters containing secondary alicyclic alcohols from chemical groups 8 and 30, and an ester of a phenol carboxylic acid from chemical group 25 - Scientific Opinion of the Panel on Food Additives, Flavourings, Processing Aids and Materials in Contact with Food View page or View pdf | |
| Flavouring Group Evaluation 9, Revision 2 (FGE.09Rev2): Secondary alicyclic saturated and unsaturated alcohols, ketones and esters containing secondary alicyclic alcohols from chemical group 8 and 30, and an ester of a phenol derivative from chemical group 25 View page or View pdf | |
| Scientific Opinion on Flavouring Group Evaluation 96 (FGE.96): Consideration of 88 flavouring substances considered by EFSA for which EU production volumes / anticipated production volumes have been submitted on request by DG SANCO. Addendum to FGE. 51, 52, 53, 54, 56, 58, 61, 62, 63, 64, 68, 69, 70, 71, 73, 76, 77, 79, 80, 83, 84, 85 and 87. View page or View pdf | |
| EPI System: | View |
| AIDS Citations: | Search |
| Cancer Citations: | Search |
| Toxicology Citations: | Search |
| EPA Substance Registry Services (TSCA): | 87061-04-9 |
| EPA ACToR: | Toxicology Data |
| EPA Substance Registry Services (SRS): | Registry |
| Laboratory Chemical Safety Summary : | 5362595 |
| National Institute of Allergy and Infectious Diseases: | Data |
| 3-(5-methyl-2-propan-2-ylcyclohexyl)oxypropane-1,2-diol | |
| Chemidplus: | 0087061049 |
References:
| Leffingwell: | Chirality or Article |
| 3-(5-methyl-2-propan-2-ylcyclohexyl)oxypropane-1,2-diol | |
| NIST Chemistry WebBook: | Search Inchi |
| Canada Domestic Sub. List: | 87061-04-9 |
| Pubchem (cid): | 5362595 |
| Pubchem (sid): | 135279774 |
Other Information:
| (IUPAC): | Atomic Weights of the Elements 2011 (pdf) |
| Videos: | The Periodic Table of Videos |
| tgsc: | Atomic Weights use for this web site |
| (IUPAC): | Periodic Table of the Elements |
| FDA Substances Added to Food (formerly EAFUS): | View |
| HMDB (The Human Metabolome Database): | HMDB36133 |
| FooDB: | FDB014982 |
| ChemSpider: | View |
| Wikipedia: | View |
Potential Blenders and core components note
Potential Uses:
| mint | FR | |
| refreshing agents |
Occurrence (nature, food, other): note
| not found in nature |
Synonyms:
| coolact 10 (Takasago) | |
| cooling agent moMOPDpd | |
| cooling agent no. 10 | |
| 3-L-(p- | menthan-3-yl oxy)-1,2-propane diol |
| 3-L-(para- | menthan-3-yl oxy)-1,2-propane diol |
| 3-L-(p- | menthan-3-yloxy)-1,2-propanediol |
| 3-L-(para- | menthan-3-yloxy)-1,2-propanediol |
| 3-L-(p- | menthane-3-yl oxy)-1,2-propane diol |
| 3-L-(para- | menthane-3-yl oxy)-1,2-propane diol |
| 3-L-(p- | menthane-3-yloxy)-1,2-propanediol |
| 3-L-(para- | menthane-3-yloxy)-1,2-propanediol |
| 3-(L- | menthoxy) propane-1,2-diol |
| 3-(L- | menthoxy)propane-1,2-diol |
| 3-1- | menthoxypropane-1,2-diol |
| 3-L- | menthoxypropane-1,2-diol |
| menthoxypropanediol | |
| 3-((L- | menthyl)oxy)propane-1,2-diol |
| 3-((5- | methyl-2-(1-methyl ethyl)cyclohexyl)oxy)-1,2-propane diol |
| 3-((5- | methyl-2-(1-methylethyl)cyclohexyl)oxy)-1,2-propandiol |
| 3-((5- | methyl-2-(1-methylethyl)cyclohexyl)oxy)-1,2-propanediol |
| 3-((5- | methyl-2-(1-methylethyl)cyclohexyl)oxy)propane-1,2-diol |
| 3-(5- | methyl-2-propan-2-ylcyclohexyl)oxypropane-1,2-diol |
| 1,2- | propanediol, 3-((5-methyl-2-(1-methylethyl)cyclohexyl)oxy)- |
| 1,2- | propanediol, 3-[[5-methyl-2-(1-methylethyl)cyclohexyl]oxy]- |
| 3-[(2-iso | propyl-5-methylcyclohexyl)oxy]propane-1,2-diol |
| rightcool cooler 10 |



