|
Category: flavor and fragrance agents
US / EU / FDA / JECFA / FEMA / FLAVIS / Scholar / Patent Information:
Physical Properties:
| Appearance: | white to yellow crystals (est) |
| Assay: | 98.00 to 100.00 %
|
| Food Chemicals Codex Listed: | No |
| Melting Point: | 44.00 to 46.00 °C. @ 760.00 mm Hg
|
| Boiling Point: | 218.00 to 219.00 °C. @ 760.00 mm Hg
|
| Vapor Pressure: | 0.083000 mmHg @ 25.00 °C. (est) |
| Vapor Density: | 4.2 ( Air = 1 ) |
| Flash Point: | 213.00 °F. TCC ( 100.56 °C. )
|
| logP (o/w): | 2.580 |
| Soluble in: |
| | water, 2346 mg/L @ 25 °C (est) | | | water, 4900 mg/L @ 25 °C (exp) |
Organoleptic Properties:
| |
| Odor Type: smoky |
| |
| Odor Strength: | high ,
recommend smelling in a 1.00 % solution or less |
| |
| Substantivity: | 109 hour(s) at 100.00 % |
| |
| | phenolic castoreum smoky guaiacol |
Odor Description:
at 1.00 % in dipropylene glycol. | phenolic castoreum smoke guaiacol
Luebke, William tgsc, (2006) |
| |
| | smoky phenolic guaiacol savory |
Odor Description:
at 1.00 %. | Smoke, phenolic, creosote and savory
Mosciano, Gerard P&F 23, No. 3, 55, (1998) |
| |
| |
| Flavor Type: smoky |
| |
| | phenolic smoky bacon ham |
Taste Description:
at 2.50 ppm. | Phenolic, smoke, bacon and ham
Mosciano, Gerard P&F 23, No. 3, 55, (1998) |
| |
| Odor and/or flavor descriptions from others (if found). |
| |
| |
Cosmetic Information:
Suppliers:
Safety Information:
| Preferred SDS: View |
| European information : |
| Most important hazard(s): | | Xi - Irritant |
R 36/37/38 - Irritating to eyes, respiratory system, and skin.
S 02 - Keep out of the reach of children.
S 24/25 - Avoid contact with skin and eyes.
S 26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S 36 - Wear suitable protective clothing.
|
| |
| Hazards identification |
| |
| Classification of the substance or mixture |
| GHS Classification in accordance with 29 CFR 1910 (OSHA HCS) |
| None found. |
| GHS Label elements, including precautionary statements |
| |
| Pictogram | |
| |
| Hazard statement(s) |
| None found. |
| Precautionary statement(s) |
| None found. |
| Oral/Parenteral Toxicity: |
oral-rat LD50 > 5000 mg/kg
(Blaszcak & Auletta, 1986)
intraperitoneal-mouse LD50 138 mg/kg
Journal of Medicinal Chemistry. Vol. 18, Pg. 868, 1975.
|
| Dermal Toxicity: |
|
Not determined
|
| Inhalation Toxicity: |
|
Not determined
|
Safety in Use Information:
| Category: | flavor and fragrance agents |
| RIFM Fragrance Material Safety Assessment: Search |
| IFRA Code of Practice Notification of the 49th Amendment to the IFRA Code of Practice |
| Recommendation for 4-ethyl phenol usage levels up to: | | | 0.0500 % in the fragrance concentrate.
|
| |
| Maximised Survey-derived Daily Intakes (MSDI-EU): | 3.50 (μg/capita/day) |
| Maximised Survey-derived Daily Intakes (MSDI-USA): | 0.10 (μg/capita/day) |
| Use levels for FEMA GRAS flavoring substances on which the FEMA Expert Panel based its judgments that the substances are generally recognized as safe (GRAS). |
| The Expert Panel also publishes separate extensive reviews of scientific information on all FEMA GRAS flavoring substances and can be found at FEMA Flavor Ingredient Library |
| publication number: 4 |
| Click here to view publication 4 |
| | average usual ppm | average maximum ppm |
| baked goods: | - | 0.20000 |
| beverages(nonalcoholic): | - | - |
| beverages(alcoholic): | - | - |
| breakfast cereal: | - | - |
| cheese: | - | - |
| chewing gum: | - | - |
| condiments / relishes: | - | - |
| confectionery froastings: | - | - |
| egg products: | - | - |
| fats / oils: | - | 0.20000 |
| fish products: | - | - |
| frozen dairy: | - | - |
| fruit ices: | - | - |
| gelatins / puddings: | - | - |
| granulated sugar: | - | - |
| gravies: | - | - |
| hard candy: | - | - |
| imitation dairy: | - | - |
| instant coffee / tea: | - | - |
| jams / jellies: | - | - |
| meat products: | - | - |
| milk products: | - | - |
| nut products: | - | - |
| other grains: | - | - |
| poultry: | - | - |
| processed fruits: | - | - |
| processed vegetables: | - | - |
| reconstituted vegetables: | - | - |
| seasonings / flavors: | - | - |
| snack foods: | - | - |
| soft candy: | - | - |
| soups: | - | - |
| sugar substitutes: | - | - |
| sweet sauces: | - | - |
Safety References:
| European Food Safety Athority(EFSA): | Flavor usage levels; Subacute, Subchronic, Chronic and Carcinogenicity Studies; Developmental / Reproductive Toxicity Studies; Genotoxicity Studies... |
| European Food Safety Authority (EFSA) reference(s): |
Opinion of the Scientific Panel on food additives, flavourings, processing aids and materials in contact with food (AFC) related to Flavouring Group Evaluation 22 (FGE.22): Ring-substituted phenolic substances from chemical groups 21 and 25 (Commission Regulation (EC) No 1565/2000 of 18 July 2000)
View page or View pdf |
Flavouring Group Evaluation 58 (FGE.58) Consideration of phenol derivatives evaluated by JECFA (55th meeting) structurally related to ring substituted phenolic substances evaluated by EFSA in FGE.22 (2006) (Commission Regulation (EC) No 1565/2000 of 18 July 2000) - Opinion of the Scientific Panel on Food Additives, Flavourings, Processing Aids and Materials in contact with Food (AFC)
View page or View pdf |
Flavouring Group Evaluation 60 (FGE.60): Consideration of eugenol and related hydroxyallylbenzene derivatives evaluated by JECFA (65th meeting) structurally related to ring- substituted phenolic substances evaluated by EFSA in FGE.22 (2006)
View page or View pdf |
Scientific Opinion on Flavouring Group Evaluation 22, Revision 1 (FGE.22Rev1): Ring-substituted phenolic substances from chemical groups 21 and 25
View page or View pdf |
Scientific Opinion on the safety and efficacy of phenol derivatives containing ring-alkyl, ring-alkoxy and side-chains with an oxygenated functional group (chemical group 25) when used as flavourings for all species
View page or View pdf |
| EPI System: | View |
| AIDS Citations: | Search |
| Cancer Citations: | Search |
| Toxicology Citations: | Search |
| EPA GENetic TOXicology: | Search |
| EPA Substance Registry Services (TSCA): | 123-07-9 |
| EPA ACToR: | Toxicology Data |
| EPA Substance Registry Services (SRS): | Registry |
| Laboratory Chemical Safety Summary : | 31242 |
| National Institute of Allergy and Infectious Diseases: | Data |
| WISER: | UN 2430 |
| WGK Germany: | 3 |
| | 4-ethylphenol |
| Chemidplus: | 0000123079 |
| RTECS: | SL4040000 for cas# 123-07-9 |
References:
Other Information:
Potential Blenders and core components note
| |
| For Odor |
| aldehydic |
| aldehydic |
| | dodecanal (aldehyde C-12 lauric) | FL/FR |
| 10- | undecenal (aldehyde C-11 undecylenic) | FL/FR |
| amber |
| | ambroxan | FL/FR |
| | angelica root oil | FL/FR |
| | cistus ladaniferus resinoid | FL/FR |
| animal |
| iso | butyl quinoline | FR |
| iso | butyl quinoline | FR |
| | costus valerolactone | FR |
| para- | cresyl caprylate | FL/FR |
| 6- | methyl quinoline | FL/FR |
| anise |
| | anise seed oil colombia | FL/FR |
| anisic |
| para- | anisaldehyde | FL/FR |
| | dihydroanethol | FL/FR |
| balsamic |
| | amyris wood oil | FL/FR |
| siam | benzoin resinoid | FL/FR |
| | benzyl salicylate | FL/FR |
| iso | bornyl acetate | FL/FR |
| laevo- | bornyl acetate | FL/FR |
| iso | butyl cinnamate | FL/FR |
| | clover nitrile | FR |
| | ethyl cinnamate | FL/FR |
| | fir balsam absolute | FR |
| | methyl cinnamate | FL/FR |
| 3- | phenyl propyl alcohol | FL/FR |
| burnt |
| | amber oil | FR |
| caramellic |
| | cyclotene | FL/FR |
| | immortelle absolute | FL/FR |
| | maltol | FL/FR |
| chocolate |
| iso | amyl phenyl acetate | FL/FR |
| | vanillyl ethyl ether | FL/FR |
| coconut |
| gamma- | heptalactone | FL/FR |
| gamma- | nonalactone (aldehyde C-18 (so-called)) | FL/FR |
| gamma- | octalactone | FL/FR |
| coffee |
| | furfuryl mercaptan | FL/FR |
| floral |
| iso | amyl salicylate | FL/FR |
| iso | butyl salicylate | FL/FR |
| | heliotropin | FL/FR |
| | heliotropyl acetone | FL/FR |
| alpha- | hexyl cinnamaldehyde | FL/FR |
| | orris pyridine 25% IPM | FR |
| | phenethyl alcohol | FL/FR |
| | phenethyl phenyl acetate | FL/FR |
| fruity |
| | ethyl 3-hydroxyhexanoate | FL/FR |
| green |
| para- | dimethyl hydroquinone | FL/FR |
| 3,7- | dimethyl-6-octenoic acid | FL/FR |
| | narcissus flower absolute | FR |
| hay |
| | hay absolute | FR |
| herbal |
| | yerba mate absolute | FL/FR |
| honey |
| | methyl phenyl acetate | FL/FR |
| leathery |
| | castoreum absolute | FL/FR |
| black | tea leaf absolute | FL/FR |
| mossy |
| | oakmoss absolute | FL/FR |
| | veramoss (IFF) | FR |
| phenolic |
| 2,3- | dimethyl benzofuran | FL/FR |
| | ethyl vanillate | FL/FR |
| ortho- | guaiacol | FL/FR |
| 4- | methyl-2,6-dimethoxyphenol | FL/FR |
| | piper betle leaf oil | FR |
| 2- | propyl phenol | FL/FR |
| 2,5- | xylenol | FL/FR |
| powdery |
| para- | anisyl acetate | FL/FR |
| roasted |
| 2- | acetyl-2-pyrroline | |
| | birch tar oil | FL/FR |
| | cade oil | FR |
| 2,6- | dimethoxyphenol | FL/FR |
| alpha- | ethoxy-ortho-cresol | FL/FR |
| | phoebe oil brazil | |
| | propyl paraben | CS |
| | pyroligneous acids | FL/FR |
| | pyroligneous acids hickory | FL/FR |
| spicy |
| | allspice oil | FL/FR |
| | ayou wood oil | FR |
| | benzyl isoeugenol | FL/FR |
| | cassia bark oil china | FL/FR |
| | clove bud oil | FL/FR |
| | cubeb oil | FL/FR |
| 4- | ethyl guaiacol | FL/FR |
| | eugenol | FL/FR |
| iso | eugenyl acetate | FL/FR |
| alpha- | methyl cinnamaldehyde | FL/FR |
| | methyl isoeugenol | FL/FR |
| | pepper tree berry oil | FL/FR |
| terpenic |
| | frankincense oil | FL/FR |
| tonka |
| | coumarin | FR |
| | tonka bean absolute | FR |
| vanilla |
| | ethyl vanillin | FL/FR |
| | vanilla bean absolute (vanilla planifolia) | FL/FR |
| | vanillyl acetate | FL/FR |
| waxy |
| | ethyl laurate | FL/FR |
| woody |
| | cistus twig/leaf oil | FL/FR |
| | guaiacwood oil | FL/FR |
| | guaiacyl acetate | FL/FR |
| 2- | methoxy-4-vinyl phenol | FL/FR |
| | methyl cedryl ketone | FL/FR |
| | patchouli ethanone | FR |
| | patchouli oil | FL/FR |
| | santall | FR |
| | tobacarol (IFF) | FR |
| | woody acetate | FR |
| (Z)- | woody amylene | FR |
| | woody propanol | FR |
| |
| For Flavor |
| |
| No flavor group found for these |
| 2- | acetyl-2-pyrroline | |
| | benzyl disulfide | FL |
| | birch tar oil | FL/FR |
| | cistus ladaniferus resinoid | FL/FR |
| alpha- | ethoxy-ortho-cresol | FL/FR |
| 1- | methyl pyrrole | FL |
| | phoebe oil brazil | |
| | prenyl mercaptan | FL |
| 2- | propyl phenol | FL/FR |
| animal |
| para- | cresyl caprylate | FL/FR |
| 6- | methyl quinoline | FL/FR |
| anise |
| | anise seed oil colombia | FL/FR |
| balsamic |
| siam | benzoin resinoid | FL/FR |
| | benzyl salicylate | FL/FR |
| laevo- | bornyl acetate | FL/FR |
| iso | butyl cinnamate | FL/FR |
| | ethyl cinnamate | FL/FR |
| berry |
| | heliotropyl acetone | FL/FR |
| burnt |
| | bacon dithiazine | FL |
| caramellic |
| | cyclotene | FL/FR |
| | maltol | FL/FR |
| chemical |
| 2,3- | dimethyl benzofuran | FL/FR |
| 2,5- | dimethyl furan | FL |
| cherry |
| | heliotropin | FL/FR |
| coconut |
| gamma- | nonalactone (aldehyde C-18 (so-called)) | FL/FR |
| coffee |
| | furfuryl mercaptan | FL/FR |
| cooling |
| iso | butyl salicylate | FL/FR |
| creamy |
| para- | anisaldehyde | FL/FR |
| | ethyl vanillate | FL/FR |
| fatty |
| bacon | fat flavor | FL |
| 10- | undecenal (aldehyde C-11 undecylenic) | FL/FR |
| floral |
| iso | amyl phenyl acetate | FL/FR |
| 3,7- | dimethyl-6-octenoic acid | FL/FR |
| | methyl phenyl acetate | FL/FR |
| | phenethyl alcohol | FL/FR |
| fruity |
| para- | anisyl acetate | FL/FR |
| | ethyl 3-hydroxyhexanoate | FL/FR |
| green |
| iso | amyl salicylate | FL/FR |
| | angelica root oil | FL/FR |
| para- | dimethyl hydroquinone | FL/FR |
| | immortelle absolute | FL/FR |
| | oakmoss absolute | FL/FR |
| herbal |
| | dihydroanethol | FL/FR |
| | yerba mate absolute | FL/FR |
| honey |
| | phenethyl phenyl acetate | FL/FR |
| lactonic |
| gamma- | heptalactone | FL/FR |
| gamma- | octalactone | FL/FR |
| leathery |
| | castoreum absolute | FL/FR |
| meaty |
| 4- | allyl-2,6-dimethoxyphenol | FL |
| medicinal |
| 2,6- | dimethoxyphenol | FL/FR |
| musty |
| 2,5- | xylenol | FL/FR |
| onion |
| 2- | methyl-1,3-dithiolane | FL |
| phenolic |
| 2- | ethyl benzene thiol | FL |
| 4- | methyl-2,6-dimethoxyphenol | FL/FR |
| roasted |
| 3,5- | diisobutyl-1,2,4-trithiolane | FL |
| rummy |
| | vanillyl ethyl ether | FL/FR |
| smoky |
| 2- | methoxy-4-vinyl phenol | FL/FR |
| | pyroligneous acids | FL/FR |
| | pyroligneous acids hickory | FL/FR |
| dextro- | xylose | FL |
| soapy |
| | dodecanal (aldehyde C-12 lauric) | FL/FR |
| spicy |
| | allspice oil | FL/FR |
| | benzyl isoeugenol | FL/FR |
| | cassia bark oil china | FL/FR |
| | chipotle chili oleoresin | FL |
| | clove bud oil | FL/FR |
| | cubeb oil | FL/FR |
| | eugenol | FL/FR |
| iso | eugenyl acetate | FL/FR |
| alpha- | methyl cinnamaldehyde | FL/FR |
| | methyl cinnamate | FL/FR |
| | methyl isoeugenol | FL/FR |
| | pepper tree berry oil | FL/FR |
| 3- | phenyl propyl alcohol | FL/FR |
| tea |
| black | tea leaf absolute | FL/FR |
| vanilla |
| | ethyl vanillin | FL/FR |
| | vanilla bean absolute (vanilla planifolia) | FL/FR |
| | vanillyl acetate | FL/FR |
| waxy |
| | ethyl laurate | FL/FR |
| alpha- | hexyl cinnamaldehyde | FL/FR |
| woody |
| | ambroxan | FL/FR |
| | amyris wood oil | FL/FR |
| iso | bornyl acetate | FL/FR |
| | cistus twig/leaf oil | FL/FR |
| 4- | ethyl guaiacol | FL/FR |
| | frankincense oil | FL/FR |
| ortho- | guaiacol | FL/FR |
| | guaiacwood oil | FL/FR |
| | guaiacyl acetate | FL/FR |
| | methyl cedryl ketone | FL/FR |
| | patchouli oil | FL/FR |
| |
Potential Uses:
Occurrence (nature, food, other): noteSynonyms:
| | benzene,1-ethyl,4-hydroxy | | p- | ethyl phenol | | para- | ethyl phenol | | 1- | ethyl-4-hydroxybenzene | | 4- | ethylphenol | | p- | ethylphenol | | para- | ethylphenol | | p- | ethylphenol (food grade) | | p- | ethylphenol natural | | 1- | hydroxy-4-ethyl benzene | | 1- | hydroxy-4-ethylbenzene | | 4- | hydroxyethyl benzene | | 4- | hydroxyethylbenzene | | 4- | hydroxyphenyl ethane | | (4- | hydroxyphenyl)ethane | | 4- | hydroxyphenylethane | | | phenol, 4-ethyl- | | | phenol, p-ethyl- |
Articles:
| US Patents: | 3,946,080 - Flavouring and perfuming ingredients |
| PubMed: | Occurrence of Brettanomyces/Dekkera in Brazilian red wines and its correlation with ethylphenols. |
| US Patents: | 3,952,024 - Furfurylthioacetone |
| PubMed: | Osmotic stress response in the wine yeast Dekkera bruxellensis. |
| PubMed: | Aroma chemistry of African Oryza glaberrima and Oryza sativa rice and their interspecific hybrids. |
| PubMed: | Comparison of fermented soybean paste (Doenjang) prepared by different methods based on profiling of volatile compounds. |
| PubMed: | [Identification of volatile organic compounds in the manures of cow, hog and chicken by solid phase microextraction coupled with gas chromatography/mass spectrometry]. |
| PubMed: | Quantitative gas chromatography-olfactometry carried out at different dilutions of an extract. Key differences in the odor profiles of four high-quality Spanish aged red wines. |
| PubMed: | A Response Surface Methodology study on the role of factors affecting growth and volatile phenol production by Brettanomyces bruxellensis ISA 2211 in wine. |
| PubMed: | Microwave-assisted deuterium exchange: the convenient preparation of isotopically labelled analogues for stable isotope dilution analysis of volatile wine phenols. |
| PubMed: | Sorption of 4-ethylguaiacol and 4-ethylphenol on yeast cell walls, using a synthetic wine. |
| PubMed: | Aromatic profile of ciders by chemical quantitative, gas chromatography-olfactometry, and sensory analysis. |
| PubMed: | Volatile constituents of commercial imported and domestic black-ripe table olives (Olea europaea). |
| PubMed: | Influence of the matrix composition on the volatility and sensory perception of 4-ethylphenol and 4-ethylguaiacol in model wine solutions. |
| PubMed: | Impact of sulfur dioxide and temperature on culturability and viability of Brettanomyces bruxellensis in Wine. |
| PubMed: | On-line monitoring of food fermentation processes using electronic noses and electronic tongues: a review. |
| PubMed: | Metabolism of nonesterified and esterified hydroxycinnamic acids in red wines by Brettanomyces bruxellensis. |
| PubMed: | Phenolic biotransformations during conversion of ferulic acid to vanillin by lactic acid bacteria. |
| PubMed: | Osmotic stress response in the wine yeast Dekkera bruxellensis. |
| PubMed: | Aroma chemistry of African Oryza glaberrima and Oryza sativa rice and their interspecific hybrids. |
| PubMed: | Biotransformation of rice bran to ferulic acid by pediococcal isolates. |
| PubMed: | Molecular and physiological comparison of spoilage wine yeasts. |
| PubMed: | Detection of Brettanomyces spp. in red wines using real-time PCR. |
| PubMed: | Effect of ethanol on the sorption of four targeted wine volatile compounds in a polyethylene film. |
| PubMed: | Characterization of the "viable but nonculturable" (VBNC) state in the wine spoilage yeast Brettanomyces. |
| PubMed: | Hydroxycinnamic acid ethyl esters as precursors to ethylphenols in wine. |
| PubMed: | Implications of Lactobacillus collinoides and Brettanomyces/Dekkera anomala in phenolic off-flavour defects of ciders. |
| PubMed: | Screening of representative cider yeasts and bacteria for volatile phenol-production ability. |
| PubMed: | Comparison of fermented soybean paste (Doenjang) prepared by different methods based on profiling of volatile compounds. |
| PubMed: | Factors influencing the production of volatile phenols by wine lactic acid bacteria. |
| PubMed: | Ustilago maydis killer toxin as a new tool for the biocontrol of the wine spoilage yeast Brettanomyces bruxellensis. |
| PubMed: | Identification and characterization of Dekkera bruxellensis, Candida pararugosa, and Pichia guilliermondii isolated from commercial red wines. |
| PubMed: | A metabolite profiling approach to identify biomarkers of flavonoid intake in humans. |
| PubMed: | Influence of the drying processes of yeasts on their volatile phenol sorption capacity in model wine. |
| PubMed: | Modeling quality of premium spanish red wines from gas chromatography-olfactometry data. |
| PubMed: | Minimization of ethylphenol precursors in red wines via the formation of pyranoanthocyanins by selected yeasts. |
| PubMed: | Molecular identification of Brettanomyces bruxellensis strains isolated from red wines and volatile phenol production. |
| PubMed: | Genetic diversity and physiological traits of Brettanomyces bruxellensis strains isolated from Tuscan Sangiovese wines. |
| PubMed: | A method for estimating Dekkera/Brettanomyces populations in wines. |
| PubMed: | Factors affecting the hydroxycinnamate decarboxylase/vinylphenol reductase activity of dekkera/brettanomyces: application for dekkera/brettanomyces control in red wine making. |
| PubMed: | Ileal and faecal digestibility of daidzein and genistein and plasma bioavailability of these isoflavones and their bioactive metabolites in the ovariectomised rat. |
| PubMed: | Effects of yeast cell-wall characteristics on 4-ethylphenol sorption capacity in model wine. |
| PubMed: | Survey of enzyme activity responsible for phenolic off-flavour production by Dekkera and Brettanomyces yeast. |
| PubMed: | Smoke-derived taint in wine: the release of smoke-derived volatile phenols during fermentation of Merlot juice following grapevine exposure to smoke. |
| PubMed: | The effect of sugar concentration and temperature on growth and volatile phenol production by Dekkera bruxellensis in wine. |
| PubMed: | Partial vinylphenol reductase purification and characterization from Brettanomyces bruxellensis. |
| PubMed: | Physiological and oenological traits of different Dekkera/Brettanomyces bruxellensis strains under wine-model conditions. |
| PubMed: | Comparison of metal oxide-based electronic nose and mass spectrometry-based electronic nose for the prediction of red wine spoilage. |
| PubMed: | Dekkera and Brettanomyces growth and utilisation of hydroxycinnamic acids in synthetic media. |
| PubMed: | Smoke-derived taint in wine: effect of postharvest smoke exposure of grapes on the chemical composition and sensory characteristics of wine. |
| PubMed: | 4-ethylphenol and 4-ethylguaiacol in wines: estimating non-microbial sourced contributions and toxicological considerations. |
| PubMed: | Multiple headspace solid-phase microextraction for eliminating matrix effect in the simultaneous determination of haloanisoles and volatile phenols in wines. |
| PubMed: | Determination of 4-ethylphenol and 4-ethylguaiacol in wines by LC-MS-MS and HPLC-DAD-fluorescence. |
| PubMed: | Phyto- and endogenous estrogens differently activate intracellular calcium ion mobilization in bovine endometrial cells. |
| PubMed: | An accurate and reproducible method for the quantitative analysis of isoflavones and their metabolites in rat plasma using liquid chromatography/mass spectrometry combined with photodiode array detection. |
| PubMed: | Sorption behavior of volatile phenols at the oak wood/wine interface in a model system. |
| PubMed: | Equol and para-ethyl-phenol stimulate prostaglandin F(2alpha) secretion in bovine corpus luteum: intracellular mechanisms of action. |
| PubMed: | Development of an enrichment medium to detect Dekkera/Brettanomyces bruxellensis, a spoilage wine yeast, on the surface of grape berries. |
| PubMed: | Phytoestrogens and their metabolites inhibit the sensitivity of the bovine corpus luteum to luteotropic factors. |
| PubMed: | Molecular typing of the yeast species Dekkera bruxellensis and Pichia guilliermondii recovered from wine related sources. |
| PubMed: | Main compounds responsible for off-odour of strawberries infected by Phytophthora cactorum. |
| PubMed: | Enrichment of cheeses manufactured from cow's and sheep's milk blends with sheep-like species-related alkylphenols. |
| PubMed: | Phenolic compounds in olive oils intended for refining: formation of 4-ethylphenol during olive paste storage. |
| PubMed: | Distribution of conjugates of alkylphenols in milk from different ruminant species. |
| PubMed: | The effects of sample preparation and gas chromatograph injection techniques on the accuracy of measuring guaiacol, 4-methylguaiacol and other volatile oak compounds in oak extracts by stable isotope dilution analyses. |
| PubMed: | Gas chromatography-olfactometry and chemical quantitative study of the aroma of six premium quality spanish aged red wines. |
| PubMed: | Volatile compounds in a spanish red wine aged in barrels made of Spanish, French, and American oak wood. |
| PubMed: | Quantitative gas chromatography-olfactometry carried out at different dilutions of an extract. Key differences in the odor profiles of four high-quality Spanish aged red wines. |
| PubMed: | Identification and quantification of impact odorants of aged red wines from Rioja. GC-olfactometry, quantitative GC-MS, and odor evaluation of HPLC fractions. |
| PubMed: | Evaluation of Smoky Taste in Cocoa Powder. |
| PubMed: | Decarboxylation of substituted cinnamic acids by lactic acid bacteria isolated during malt whisky fermentation. |
|
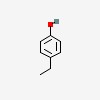 3D/inchi
3D/inchi











