Articles:
methylsulfanylbenzene
Notes:
Found in coffee. Flavouring agent for baked and meat products and seaonings. Food additive listed in the Food Additive Database (Jan. 2001)
| CAS Number: | 100-68-5 | 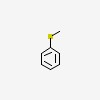 3D/inchi 3D/inchi
|
| ECHA EINECS - REACH Pre-Reg: | 202-878-2 | |
| FDA UNII: | BB4K737YF4 | |
| Nikkaji Web: | J3.592K | |
| Beilstein Number: | 1904179 | |
| MDL: | MFCD00008559 | |
| CoE Number: | 11533 | |
| XlogP3: | 2.70 (est) | |
| Molecular Weight: | 124.20576000 | |
| Formula: | C7 H8 S | |
| NMR Predictor: | Predict (works with chrome or firefox) | |
Category: flavoring agents
US / EU / FDA / JECFA / FEMA / FLAVIS / Scholar / Patent Information:
| Google Scholar: | Search |
| Google Books: | Search |
| Google Scholar: with word "volatile" | Search |
| Google Scholar: with word "flavor" | Search |
| Google Scholar: with word "odor" | Search |
| Perfumer and Flavorist: | Search |
| Google Patents: | Search |
| US Patents: | Search |
| EU Patents: | Search |
| Pubchem Patents: | Search |
| PubMed: | Search |
| NCBI: | Search |
| JECFA Food Flavoring: | 459 methyl phenyl sulfide |
| DG SANTE Food Flavourings: | 12.162 methyl phenyl sulfide |
| FEMA Number: | 3873 methyl phenyl sulfide |
| FDA: | No longer provide for the use of these seven synthetic flavoring substances |
| FDA Mainterm (SATF): | 100-68-5 ; METHYL PHENYL SULFIDE |
Physical Properties:
| Appearance: | colorless to pale yellow clear liquid (est) |
| Assay: | 98.00 to 100.00 % |
| Food Chemicals Codex Listed: | No |
| Specific Gravity: | 0.95800 to 0.96800 @ 25.00 °C. |
| Pounds per Gallon - (est).: | 7.972 to 8.055 |
| Refractive Index: | 1.53200 to 1.55100 @ 20.00 °C. |
| Melting Point: | -15.00 °C. @ 760.00 mm Hg |
| Boiling Point: | 188.00 to 193.00 °C. @ 760.00 mm Hg |
| Vapor Pressure: | 0.662000 mmHg @ 25.00 °C. (est) |
| Flash Point: | 135.00 °F. TCC ( 57.22 °C. ) |
| logP (o/w): | 2.740 |
| Soluble in: | |
| alcohol | |
| oils | |
| water, 442.6 mg/L @ 25 °C (est) | |
| water, 506 mg/L @ 25 °C (exp) | |
| Insoluble in: | |
| water | |
Organoleptic Properties:
| Odor Type: solvent | |
| solvent spicy woody sawdust | |
| Odor Description: at 0.10 % in propylene glycol. | toluene solvent spicy woody sawdust |
| solvent spicy pungent woody sawdust | |
| Odor Description: at 0.10 % in ethyl alcohol. | Toluene solvent, spicy, pungent, woody sawdust Mosciano, Gerard P&F 24, No. 6, 10, (1999) |
| Flavor Type: solvent | |
| solvent woody coffee roasted coffee | |
| Taste Description: at 0.02 - 0.50 ppm. | Solventy, woody, roasted coffee Mosciano, Gerard P&F 24, No. 6, 10, (1999) |
| Odor and/or flavor descriptions from others (if found). | |
Cosmetic Information:
| None found |
Suppliers:
| Beijing Lys Chemicals |
| Methyl Phenyl Sulfide |
| BOC Sciences |
| For experimental / research use only. |
| Thioanisole |
| EMD Millipore |
| For experimental / research use only. |
| Methyl Phenyl Sulfide |
| Parchem |
| methyl phenyl sulfide |
| Penta International |
| METHYL PHENYL SULFIDE |
| Santa Cruz Biotechnology |
| For experimental / research use only. |
| Thioanisole |
| Sigma-Aldrich |
| For experimental / research use only. |
| Thioanisole analytical standard |
| TCI AMERICA |
| For experimental / research use only. |
| Thioanisole >99.0%(GC) |
| Tengzhou Jitian Aroma Chemiclal |
| Methyl Phenyl Sulfide |
Safety Information:
| Preferred SDS: View | |
| European information : | |
| Most important hazard(s): | |
| Xn - Harmful. | |
|
R 22 - Harmful if swallowed. R 36/37/38 - Irritating to eyes, respiratory system, and skin. S 02 - Keep out of the reach of children. S 20/21 - When using do not eat, drink or smoke. S 24/25 - Avoid contact with skin and eyes. S 26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S 36/37/39 - Wear suitable clothing, gloves and eye/face protection. | |
| Hazards identification | |
| Classification of the substance or mixture | |
| GHS Classification in accordance with 29 CFR 1910 (OSHA HCS) | |
| None found. | |
| GHS Label elements, including precautionary statements | |
| Pictogram | |
| Hazard statement(s) | |
| None found. | |
| Precautionary statement(s) | |
| None found. | |
| Oral/Parenteral Toxicity: | |
|
intravenous-mouse LD50 56 mg/kg U.S. Army Armament Research & Development Command, Chemical Systems Laboratory, NIOSH Exchange Chemicals. Vol. NX#00184 oral-rat LD50 891 mg/kg Pesticide Biochemistry and Physiology. Vol. 1, Pg. 356, 1971. | |
| Dermal Toxicity: | |
| Not determined | |
| Inhalation Toxicity: | |
| Not determined | |
Safety in Use Information:
| Category: | flavoring agents | ||
| Recommendation for methyl phenyl sulfide usage levels up to: | |||
| not for fragrance use. | |||
| Maximised Survey-derived Daily Intakes (MSDI-EU): | 0.012 (μg/capita/day) | ||
| Maximised Survey-derived Daily Intakes (MSDI-USA): | 0.40 (μg/capita/day) | ||
| Modified Theoretical Added Maximum Daily Intake (mTAMDI): | ND (μg/person/day) | ||
| Threshold of Concern: | 540 (μg/person/day) | ||
| Structure Class: | II | ||
| Use levels for FEMA GRAS flavoring substances on which the FEMA Expert Panel based its judgments that the substances are generally recognized as safe (GRAS). | |||
| The Expert Panel also publishes separate extensive reviews of scientific information on all FEMA GRAS flavoring substances and can be found at FEMA Flavor Ingredient Library | |||
| publication number: 18 | |||
| Click here to view publication 18 | |||
| average usual ppm | average maximum ppm | ||
| baked goods: | 0.06000 | 1.00000 | |
| beverages(nonalcoholic): | 0.20000 | 2.00000 | |
| beverages(alcoholic): | - | - | |
| breakfast cereal: | - | - | |
| cheese: | - | - | |
| chewing gum: | - | - | |
| condiments / relishes: | - | - | |
| confectionery froastings: | - | - | |
| egg products: | - | - | |
| fats / oils: | 0.01000 | 0.04000 | |
| fish products: | - | - | |
| frozen dairy: | - | - | |
| fruit ices: | - | - | |
| gelatins / puddings: | 0.10000 | 1.00000 | |
| granulated sugar: | - | - | |
| gravies: | 0.10000 | 1.00000 | |
| hard candy: | - | - | |
| imitation dairy: | - | - | |
| instant coffee / tea: | - | - | |
| jams / jellies: | - | - | |
| meat products: | 0.40000 | 4.00000 | |
| milk products: | - | - | |
| nut products: | - | - | |
| other grains: | - | - | |
| poultry: | - | - | |
| processed fruits: | - | - | |
| processed vegetables: | - | - | |
| reconstituted vegetables: | - | - | |
| seasonings / flavors: | 0.50000 | 1.00000 | |
| snack foods: | - | - | |
| soft candy: | - | - | |
| soups: | - | - | |
| sugar substitutes: | - | - | |
| sweet sauces: | - | - | |
Safety References:
| European Food Safety Athority(EFSA): | Flavor usage levels; Subacute, Subchronic, Chronic and Carcinogenicity Studies; Developmental / Reproductive Toxicity Studies; Genotoxicity Studies... |
| European Food Safety Authority (EFSA) reference(s): | |
| Flavouring Group Evaluation 8 (FGE.08)[1]: Aliphatic and alicyclic mono-, di-, tri-, and polysulphides with or without additional oxygenated functional groups from chemical group 20 View page or View pdf | |
| Flavouring Group Evaluation 8, Revision 1 (FGE.08Rev1): Aliphatic and alicyclic mono-, di-, tri-, and polysulphides with or without additional oxygenated functional groups from chemical groups 20 and 30 View page or View pdf | |
| Scientific Opinion on Flavouring Group Evaluation 8, Revision 3 (FGE.08Rev3): Aliphatic and alicyclic mono-, di-, tri-, and polysulphides with or without additional oxygenated functional groups from chemical groups 20 and 30 View page or View pdf | |
| Scientific Opinion on Flavouring Group Evaluation 91, Revision 1 (FGE.91Rev1): Consideration of simple aliphatic and aromatic sulphides and thiols evaluated by JECFA (53rd and 68th meetings) structurally related to aliphatic and alicyclic mono-, di-, tri-, and polysulphides with or without additional oxygenated functional groups evaluated by EFSA in FGE.08Rev3 (2011) View page or View pdf | |
| Scientific Opinion on Flavouring Group Evaluation 08, Revision 4 (FGE.08Rev4): Aliphatic and alicyclic mono-, di-, tri-, and polysulphides with or without additional oxygenated functional groups from chemical groups 20 and 30 View page or View pdf | |
| Scientific Opinion on Flavouring Group Evaluation 91, Revision 2 (FGE.91Rev2): Consideration of simple aliphatic and aromatic sulphides and thiols evaluated by the JECFA (53rd and 68th meetings) structurally related to aliphatic and alicyclic mono-, di-, tri-, and polysulphides with or without additional oxygenated functional groups evaluated by EFSA in FGE.08Rev5 (2012) View page or View pdf | |
| Scientific Opinion on Flavouring Group Evaluation 91, Revision 3 (FGE.91Rev3): consideration of aliphatic, aromatic and a,�-unsaturated sulfides and thiols evaluated by JECFA (53rd, 61st, 68th and 76th meetings), structurally related to substances in FGE.08Rev5 View page or View pdf | |
| EPI System: | View |
| AIDS Citations: | Search |
| Cancer Citations: | Search |
| Toxicology Citations: | Search |
| EPA Substance Registry Services (TSCA): | 100-68-5 |
| EPA ACToR: | Toxicology Data |
| EPA Substance Registry Services (SRS): | Registry |
| Laboratory Chemical Safety Summary : | 7520 |
| National Institute of Allergy and Infectious Diseases: | Data |
| WGK Germany: | 3 |
| methylsulfanylbenzene | |
| Chemidplus: | 0000100685 |
| RTECS: | DA6200000 for cas# 100-68-5 |
References:
| methylsulfanylbenzene | |
| NIST Chemistry WebBook: | Search Inchi |
| Pubchem (cid): | 7520 |
| Pubchem (sid): | 134972860 |
Other Information:
| (IUPAC): | Atomic Weights of the Elements 2011 (pdf) |
| Videos: | The Periodic Table of Videos |
| tgsc: | Atomic Weights use for this web site |
| (IUPAC): | Periodic Table of the Elements |
| FDA Substances Added to Food (formerly EAFUS): | View |
| CHEMBL: | View |
| HMDB (The Human Metabolome Database): | HMDB34448 |
| FooDB: | FDB012855 |
| Export Tariff Code: | 2930.90.2900 |
| VCF-Online: | VCF Volatile Compounds in Food |
| ChemSpider: | View |
| FAO: | Methyl phenyl sulfide |
Potential Blenders and core components note
Potential Uses:
| cherry | FL | |
| mint | FL | |
| timber | ||
| whiskey | FL | |
| woody | FR |
Occurrence (nature, food, other): note
| coffee Search PMC Picture |
Synonyms:
| benzene, (methylthio)- | |
| methyl phenyl sulphide | |
| methyl phenyl thioether | |
| methyl phenylsulfide | |
| ( | methyl thio) benzene |
| methyl-phenyl sulfide | |
| methylphenylsulfide | |
| ( | methylsulfanyl)benzene |
| methylsulfanylbenzene | |
| ( | methylthio)benzene |
| phenyl methyl sulfide | |
| phenyl thiomethane | |
| 1- | phenyl-1-thiaethane |
| 1- | phenyl-1-thioethane |
| phenylthiomethane | |
| sulfide, methyl phenyl | |
| (1- | thiaethyl) benzene |
| (1- | thiaethyl)benzene |
| thioanisol | |
| thioanisole |



