|
Category: flavor and fragrance agents
US / EU / FDA / JECFA / FEMA / FLAVIS / Scholar / Patent Information:
Physical Properties:
| Appearance: | colorless clear liquid (est) |
| Assay: | 95.00 to 100.00 % sum of isomers
|
| Food Chemicals Codex Listed: | No |
| Specific Gravity: | 0.84700 to 0.85800 @ 25.00 °C.
|
| Pounds per Gallon - (est).: | 7.048 to 7.139
|
| Refractive Index: | 1.46000 to 1.46600 @ 20.00 °C.
|
| Boiling Point: | 202.00 to 204.00 °C. @ 10.00 mm Hg
|
| Boiling Point: | 333.00 to 335.00 °C. @ 760.00 mm Hg
|
| Vapor Pressure: | 0.000008 mmHg @ 25.00 °C. (est) |
| Flash Point: | > 212.00 °F. TCC ( > 100.00 °C. )
|
| logP (o/w): | 8.230 (est) |
| Soluble in: |
| | water, 0.00327 mg/L @ 25 °C (est) |
Organoleptic Properties:
| |
| Odor Type: floral |
| |
| Odor Strength: | low |
| |
| | floral balsamic powdery waxy |
Odor Description:
at 100.00 %. | delicate floral balsam powdery waxy
Luebke, William tgsc, (1987) |
| |
| |
| Flavor Type: floral |
| |
| | floral balsamic green berry tropical vegetable waxy |
Taste Description:
| mild floral balsamic green berry tropical vegetable waxy
Luebke, William tgsc, (1987) |
| |
| Odor and/or flavor descriptions from others (if found). |
| |
| |
Cosmetic Information:
Suppliers:
Safety Information:
| European information : |
| Most important hazard(s): | | None - None found. |
S 02 - Keep out of the reach of children.
S 24/25 - Avoid contact with skin and eyes.
S 36 - Wear suitable protective clothing.
|
| |
| Hazards identification |
| |
| Classification of the substance or mixture |
| GHS Classification in accordance with 29 CFR 1910 (OSHA HCS) |
| None found. |
| GHS Label elements, including precautionary statements |
| |
| Pictogram | |
| |
| Hazard statement(s) |
| None found. |
| Precautionary statement(s) |
| None found. |
| Oral/Parenteral Toxicity: |
oral-rat LD50 > 5000 mg/kg
Food and Chemical Toxicology. Vol. 20, Pg. 811, 1982.
|
| Dermal Toxicity: |
skin-rabbit LD50 > 5000 mg/kg
Food and Chemical Toxicology. Vol. 20, Pg. 811, 1982.
|
| Inhalation Toxicity: |
|
Not determined
|
Safety in Use Information:
| Category: | flavor and fragrance agents |
| RIFM Fragrance Material Safety Assessment: Search |
| IFRA Code of Practice Notification of the 49th Amendment to the IFRA Code of Practice |
| Recommendation for phytol usage levels up to: | | | 10.0000 % in the fragrance concentrate.
|
| |
| Maximised Survey-derived Daily Intakes (MSDI-EU): | 0.32 (μg/capita/day) |
| Maximised Survey-derived Daily Intakes (MSDI-USA): | ND (μg/capita/day) |
| Modified Theoretical Added Maximum Daily Intake (mTAMDI): | 2300 (μg/person/day) |
| Threshold of Concern: | 1800 (μg/person/day) |
| Structure Class: | I |
| Use levels for FEMA GRAS flavoring substances on which the FEMA Expert Panel based its judgments that the substances are generally recognized as safe (GRAS). |
| The Expert Panel also publishes separate extensive reviews of scientific information on all FEMA GRAS flavoring substances and can be found at FEMA Flavor Ingredient Library |
| publication number: 22 |
| Click here to view publication 22 |
| | average usual ppm | average maximum ppm |
| baked goods: | 10.00000 | 50.00000 |
| beverages(nonalcoholic): | 5.00000 | 25.00000 |
| beverages(alcoholic): | - | - |
| breakfast cereal: | 5.00000 | 25.00000 |
| cheese: | 7.00000 | 35.00000 |
| chewing gum: | - | - |
| condiments / relishes: | 5.00000 | 25.00000 |
| confectionery froastings: | 10.00000 | 50.00000 |
| egg products: | - | - |
| fats / oils: | 5.00000 | 25.00000 |
| fish products: | 2.00000 | 10.00000 |
| frozen dairy: | 7.00000 | 35.00000 |
| fruit ices: | 10.00000 | 50.00000 |
| gelatins / puddings: | 5.00000 | 25.00000 |
| granulated sugar: | - | - |
| gravies: | 20.00000 | 100.00000 |
| hard candy: | - | - |
| imitation dairy: | 7.00000 | 35.00000 |
| instant coffee / tea: | - | - |
| jams / jellies: | 5.00000 | 25.00000 |
| meat products: | 2.00000 | 10.00000 |
| milk products: | 7.00000 | 35.00000 |
| nut products: | - | - |
| other grains: | 5.00000 | 25.00000 |
| poultry: | 2.00000 | 10.00000 |
| processed fruits: | 7.00000 | 35.00000 |
| processed vegetables: | - | - |
| reconstituted vegetables: | - | - |
| seasonings / flavors: | 5.00000 | 25.00000 |
| snack foods: | 10.00000 | 50.00000 |
| soft candy: | - | - |
| soups: | 5.00000 | 25.00000 |
| sugar substitutes: | - | - |
| sweet sauces: | 5.00000 | 25.00000 |
| |
| Food categories according to Commission Regulation EC No. 1565/2000 (EC, 2000) in FGE.06 (EFSA, 2002a). According to the Industry the "normal" use is defined as the average of reported usages and "maximum use" is defined as the 95th percentile of reported usages (EFSA, 2002i). |
| Note: mg/kg = 0.001/1000 = 0.000001 = 1/1000000 = ppm. |
| | average usage mg/kg | maximum usage mg/kg |
| Dairy products, excluding products of category 02.0 (01.0): | 7.00000 | 35.00000 |
| Fats and oils, and fat emulsions (type water-in-oil) (02.0): | 5.00000 | 25.00000 |
| Edible ices, including sherbet and sorbet (03.0): | 10.00000 | 50.00000 |
| Processed fruit (04.1): | 7.00000 | 35.00000 |
| Processed vegetables (incl. mushrooms & fungi, roots & tubers, pulses and legumes), and nuts & seeds (04.2): | - | - |
| Confectionery (05.0): | 10.00000 | 50.00000 |
| Chewing gum (05.0): | - | - |
| Cereals and cereal products, incl. flours & starches from roots & tubers, pulses & legumes, excluding bakery (06.0): | 5.00000 | 25.00000 |
| Bakery wares (07.0): | 10.00000 | 50.00000 |
| Meat and meat products, including poultry and game (08.0): | 2.00000 | 10.00000 |
| Fish and fish products, including molluscs, crustaceans and echinoderms (MCE) (09.0): | 2.00000 | 10.00000 |
| Eggs and egg products (10.0): | - | - |
| Sweeteners, including honey (11.0): | - | - |
| Salts, spices, soups, sauces, salads, protein products, etc. (12.0): | 5.00000 | 25.00000 |
| Foodstuffs intended for particular nutritional uses (13.0): | 10.00000 | 50.00000 |
| Non-alcoholic ("soft") beverages, excl. dairy products (14.1): | - | - |
| Alcoholic beverages, incl. alcohol-free and low-alcoholic counterparts (14.2): | 10.00000 | 50.00000 |
| Ready-to-eat savouries (15.0): | 20.00000 | 100.00000 |
| Composite foods (e.g. casseroles, meat pies, mincemeat) - foods that could not be placed in categories 01.0 - 15.0 (16.0): | 5.00000 | 25.00000 |
Safety References:
| European Food Safety Athority(EFSA): | Flavor usage levels; Subacute, Subchronic, Chronic and Carcinogenicity Studies; Developmental / Reproductive Toxicity Studies; Genotoxicity Studies... |
| European Food Safety Authority (EFSA) reference(s): |
Flavouring Group Evaluation 202: 3-Alkylated aliphatic acyclic alpha,beta-unsaturated aldehydes and precursors with or without additional double bonds from chemical subgroup 1.1.3 of FGE.19[1]
View page or View pdf |
Scientific Opinion on Flavouring Group Evaluation 95 (FGE.95): Consideration of aliphatic, linear or branched-chain saturated and unsaturated alcohols, aldehydes, acids and related esters evaluated by JECFA (69th meeting) structurally related to esters of branched- and straight-chain aliphatic saturated primary alcohols and of one secondary alcohol, and branched- and straight-chain unsaturated carboxylic acids evaluated by EFSA in FGE.05Rev1 (2008)
View page or View pdf |
| EPI System: | View |
| Chemical Carcinogenesis Research Information System: | Search |
| AIDS Citations: | Search |
| Cancer Citations: | Search |
| Toxicology Citations: | Search |
| EPA Substance Registry Services (TSCA): | 150-86-7 |
| EPA ACToR: | Toxicology Data |
| EPA Substance Registry Services (SRS): | Registry |
| Laboratory Chemical Safety Summary : | 5280435 |
| National Institute of Allergy and Infectious Diseases: | Data |
| WGK Germany: | 2 |
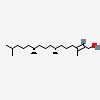
| | (E,7R,11R)-3,7,11,15-tetramethylhexadec-2-en-1-ol |
| Chemidplus: | 0000150867 |
| RTECS: | TJ3490000 for cas# 150-86-7 |
References:
Other Information:
Potential Blenders and core components notePotential Uses:
Occurrence (nature, food, other): noteSynonyms:
| 2- | hexadecen-1-ol, 3,7,11,15-tetramethyl-, (2E,7R,11R)- | | 2- | hexadecen-1-ol, 3,7,11,15-tetramethyl-, (R-(R*,R*-(E)))- | | (E,Z)- | phytol | | (E)- | phytol | | trans- | phytol | | | phytol natural | | 3,7,11,15- | tetramethyl hexadec-2-en-1-ol | | (2E,7R,11R)- 3,7,11,15- | tetramethyl-2-hexadecen-1-ol | | (2E,7R,11R)-3,7,11,15- | tetramethyl-2-hexadecen-1-ol | | (R-(R*,R* -(E)))-3,7,11,15- | tetramethyl-2-hexadecen-1-ol | | (theta-(theta, theta-(E)))-3,7,11,15- | tetramethyl-2-hexadecen-1-ol | | (2E,7R,11R)-3,7,11,15- | tetramethylhexadec-2-en-1-ol | | (2E)(7R,11R)-3,7,11,15- | tetramethylhexadec-2-en-1-ol | | (E,7R,11R)-3,7,11,15- | tetramethylhexadec-2-en-1-ol |
Articles:
| PubMed: | Purification and Immobilization of the Recombinant Brassica oleracea Chlorophyllase 1 (BoCLH1) on DIAION®CR11 as Potential Biocatalyst for the Production of Chlorophyllide and Phytol. |
| PubMed: | A novel ether-linked phytol-containing digalactosylglycerolipid in the marine green alga, Ulva pertusa. |
| PubMed: | In vitro anti-quorum sensing activity of phytol. |
| PubMed: | Chlorophyll degradation: the tocopherol biosynthesis-related phytol hydrolase in Arabidopsis seeds is still missing. |
| PubMed: | An insight into the cytotoxic activity of phytol at in vitro conditions. |
| PubMed: | Phytol derivatives as drug resistance reversal agents. |
| PubMed: | Two natural products, trans-phytol and (22E)-ergosta-6,9,22-triene-3β,5α,8α-triol, inhibit the biosynthesis of estrogen in human ovarian granulosa cells by aromatase (CYP19). |
| PubMed: | Further in vitro evaluation of antiradical and antimicrobial activities of phytol. |
| PubMed: | Phytol, a diterpene alcohol from chlorophyll, as a drug against neglected tropical disease Schistosomiasis mansoni. |
| PubMed: | Anxiolytic-like effects of phytol: possible involvement of GABAergic transmission. |
| PubMed: | Mitsunobu reactions of 5-fluorouridine with the terpenols phytol and nerol: DNA building blocks for a biomimetic lipophilization of nucleic acids. |
| PubMed: | Phytol, a diterpene alcohol, inhibits the inflammatory response by reducing cytokine production and oxidative stress. |
| PubMed: | Effect of dietary phytol on the expression of α-amino-β-carboxymuconate-ε-semialdehyde decarboxylase, a key enzyme of tryptophan-niacin metabolism, in rats. |
| PubMed: | Efficacy of phytol-derived diterpenoid immunoadjuvants over alum in shaping the murine host's immune response to Staphylococcus aureus. |
| PubMed: | Phytol/Phytanic acid and insulin resistance: potential role of phytanic acid proven by docking simulation and modulation of biochemical alterations. |
| PubMed: | Synthetic adjuvants for vaccine formulations: phytol derivatives. |
| PubMed: | Anticonvulsant effect of phytol in a pilocarpine model in mice. |
| PubMed: | Differentiation of refined and virgin edible oils by means of the trans- and cis-phytol isomer distribution. |
| PubMed: | Phytol-derived novel isoprenoid immunostimulants. |
| PubMed: | Synthetic adjuvants for vaccine formulations: evaluation of new phytol derivatives in induction and persistence of specific immune response. |
| PubMed: | Molecular signatures of phytol-derived immunostimulants in the context of chemokine-cytokine microenvironment and enhanced immune response. |
| PubMed: | Anti-scratching behavioral effect of the essential oil and phytol isolated from Artemisia princeps Pamp. in mice. |
| PubMed: | Fragrance material review on phytol. |
| PubMed: | Antitubercular potential of some semisynthetic analogues of phytol. |
| PubMed: | Phytol-induced hepatotoxicity in mice. |
| PubMed: | Continuous chlorophyll degradation accompanied by chlorophyllide and phytol reutilization for chlorophyll synthesis in Synechocystis sp. PCC 6803. |
| PubMed: | Phytol-based novel adjuvants in vaccine formulation: 1. assessment of safety and efficacy during stimulation of humoral and cell-mediated immune responses. |
| PubMed: | Phytol-based novel adjuvants in vaccine formulation: 2. Assessment of efficacy in the induction of protective immune responses to lethal bacterial infections in mice. |
| PubMed: | Metabolism of phytol to phytanic acid in the mouse, and the role of PPARalpha in its regulation. |
| PubMed: | Phytanic acid: production from phytol, its breakdown and role in human disease. |
| PubMed: | Polyunsaturated fats attenuate the dietary phytol-induced increase in hepatic fatty acid oxidation in mice. |
| PubMed: | Peroxisomal trans-2-enoyl-CoA reductase is involved in phytol degradation. |
| PubMed: | The Arabidopsis vitamin E pathway gene5-1 mutant reveals a critical role for phytol kinase in seed tocopherol biosynthesis. |
| PubMed: | A salvage pathway for phytol metabolism in Arabidopsis. |
| PubMed: | Phytol as one of the determinants of chlorophyll interactions in solution. |
| PubMed: | Phytol directly activates peroxisome proliferator-activated receptor alpha (PPARalpha) and regulates gene expression involved in lipid metabolism in PPARalpha-expressing HepG2 hepatocytes. |
| PubMed: | Phytol-modified heme in mesoporous silica: conjugates as models of hemoproteins. |
| PubMed: | Evidence for the preventive effect of the polyunsaturated phytol side chain in tocotrienols on 17beta-estradiol epoxidation. |
| PubMed: | Characterization of the final step in the conversion of phytol into phytanic acid. |
| PubMed: | Biphasic effects of geranylgeraniol, teprenone, and phytol on the growth of Staphylococcus aureus. |
| PubMed: | A phytol-enriched diet induces changes in fatty acid metabolism in mice both via PPARalpha-dependent and -independent pathways. |
| PubMed: | Assay for Sjögren-Larsson syndrome based on a deficiency of phytol degradation. |
| PubMed: | Clinical significance of oxidation from phytol to phytanic acid in man. |
| PubMed: | Potentiation of the teratogenic effects induced by coadministration of retinoic acid or phytanic acid/phytol with synthetic retinoid receptor ligands. |
| PubMed: | The methylerythritol phosphate pathway contributes to carotenoid but not phytol biosynthesis in Euglena gracilis. |
| PubMed: | Identification of fatty aldehyde dehydrogenase in the breakdown of phytol to phytanic acid. |
| PubMed: | Simulated moving bed chromatography with supercritical fluids for the resolution of bi-naphthol enantiomers and phytol isomers. |
| PubMed: | Phytol, SSADH inhibitory diterpenoid of Lactuca sativa. |
| PubMed: | Diol- and triol-types of phytol induce apoptosis in lymphoid leukemia Molt 4B cells. |
| PubMed: | Phytanic acid, but not pristanic acid, mediates the positive effects of phytol derivatives on brown adipocyte differentiation. |
| PubMed: | Prevention of vitamin A teratogenesis by phytol or phytanic acid results from reduced metabolism of retinol to the teratogenic metabolite, all-trans-retinoic acid. |
| PubMed: | A convenient and asymmetric protocol for the synthesis of natural products containing chiral alkyl chains via Zr-catalyzed asymmetric carboalumination of alkenes. Synthesis of phytol and vitamins E and K. |
| PubMed: | Limonoids and phytol derivatives from Cedrela sinensis. |
| PubMed: | Biodegradation of free phytol by bacterial communities isolated from marine sediments under aerobic and denitrifying conditions. |
| PubMed: | Phytol induces programmed cell death in human lymphoid leukemia Molt 4B cells. |
| PubMed: | Phytol is a novel tumor promoter on ICR mouse skin. |
| PubMed: | Biosynthesis of phytol in the cyanobacterium synechocystis sp. UTEX 2470: utilization of the non-mevalonate pathway |
| PubMed: | Antimycobacterial activity of (E)-phytol and derivatives: a preliminary structure-activity study. |
| PubMed: | Incorporation of 1-deoxy-D-xylulose into isoprene and phytol by higher plants and algae. |
| PubMed: | Phytol metabolites are circulating dietary factors that activate the nuclear receptor RXR. |
| PubMed: | Antispasmodic activity of beta-damascenone and E-phytol isolated from Ipomoea pes-caprae. |
| PubMed: | Isolation of trans-Phytol from Phyllanthus niruri1. |
| PubMed: | Deuterium NMR study of the interaction of phytanic acid and phytol with the head group region of a phospholipid bilayer. Evidence of magnetic orientation. |
| PubMed: | Free radical damage in neonatal rat cardiac myocyte cultures: effects of alpha-tocopherol, Trolox, and phytol. |
| PubMed: | Deuterium nuclear magnetic resonance study of the interaction of branched chain compounds (phytanic acid, phytol) with a phospholipid model membrane. |
| PubMed: | Characterization of phytol-phytanate conversion activity in rat liver. |
| PubMed: | Phytol and peroxisome proliferation. |
| PubMed: | In vitro conversion of phytol to phytanic acid in rat liver: subcellular distribution of activity and chemical characterization of intermediates using a new bromination technique. |
| PubMed: | The effect of phytol upon skeletal muscle damage in vitamin E-deficient animals. |
| PubMed: | Changes in sterol and phospholipid fatty acid composition in Refsum's disease fibroblasts grown in the presence of phytol. |
| PubMed: | 3-Methyladipate excretion in animals fed a phytol supplement with reference to Refsum's disease. |
| PubMed: | Incorporation of phytol precursors into chlorophylls of tobacco cell cultures. |
| PubMed: | Terminal steps of bacteriochlorophyll a phytol formation in purple photosynthetic bacteria. |
| PubMed: | Determination of pheophorbide a, pyropheophorbide a and phytol. |
| PubMed: | Effects of phytol, a branched, long-chain aliphatic alcohol, on biochemical values and on hepatic peroxisomal enzymes of rats. |
| PubMed: | Phytol degradation by marine bacteria. |
| PubMed: | [Refsum's disease. 14 years of a diet without phytol]. |
| PubMed: | [Refsum's disease: 10 years of a diet low in phytanic acid and phytol]. |
| PubMed: | Phytol synthesis from geranylgeraniol in spinach chloroplasts. |
| PubMed: | Refsum's disease. Eye manifestations in a patient treated with low phytol low phytanic acid diet. |
| PubMed: | Acyclic diterpene alcohols: occurrence and synthesis of geranylcitronellol, phytol and geranylgeraniol. |
| PubMed: | [Dietary treatment of a patient with Refsum's syndrome (clinical and chemical course observation during a phytol and phytanic acid-poor diet)]. |
| PubMed: | Characterization of bovine rumen liquor isoprenoid hydrocarbons with reference to dietary phytol. |
| PubMed: | [Light dependence of phytol accumulation. A contribution to the question of chlorophyll biosynthesis]. |
| PubMed: | Metabolism of long-chain isoprenoid alcohols. Incorporation of phytol and dihydrophytol into the lipids of rat brain. |
| PubMed: | [Occurrence of phytol and geranylgeraniol in the bacteriochlorophylls of red and green sulfur bacteria (author's transl)]. |
| PubMed: | Factors affecting the formation of phytol and its incorporation into chlorophyll by homogenates of the leaves of the french bean Phaseolus vulgaris. |
| PubMed: | Synthesis and acid hydrolysis of monophosphate and pyrophosphate esters of phytanol and phytol. |
| PubMed: | [Refsum's disease. Phytol loading test in an offspring]. |
| PubMed: | [Method for the identification and quantitative determination of phytol]. |
| PubMed: | [Phytol and phytanic acid in 2 cases of hereditary chronic polyneuropathy with uncertain classification: correlations with Refsum's disease]. |
| PubMed: | Phytol and Bacteriochlorophyll Synthesis in Rhodopseudomonas spheroides. |
| PubMed: | Fate of 14 C-phytol administered orally to sheep. |
| PubMed: | Effect of phytol on the incidence of muscular dystrophy in rabbits. |
| PubMed: | [Phytol intolerance: Refsum's disease]. |
| PubMed: | Effects of dietary phytol on tissue and plasma lipids in the rabbit. Biosynthesis of glyceryl-triphytanate in the heart. |
| PubMed: | Increased incidence of encephalomalacia in the chick by dietary phytol. |
| PubMed: | [Phytol in yellowed leaves]. |
| PubMed: | Biosynthesis of glyceryl triphytanate in rabbit receiving a diet enriche with phytol. |
| PubMed: | Phytol-derived C19 di- and triolefinic hydrocarbons in marine zooplankton and fishes. |
| PubMed: | A bacterium capable of using phytol as its sole carbon source, isolated from algal sediment of Mud Lake, Florida. |
| PubMed: | Phytenic acid: identification of five isomers in chemical and biological products of phytol. |
| PubMed: | [Role of phytanic acid and phytol in fish with avitaminosis E]. |
| PubMed: | [Refsum's disease. Clinical and biochemical evolution under a phytol-free diet. Complementary biochemical studies. Supplement of the 1966 report]. |
| PubMed: | Absorption of chlorophyll phytol in normal man and in patients with Refsum's disease. |
| PubMed: | Absorption of phytol from dietary chlorophyll in the rat. |
| PubMed: | Incorporation of DL-[2-14C]mevalonic acid lactone into beta-carotene and the phytol side chain of chlorophyll in cotyledons of four species of pine seedlings. |
| PubMed: | [Influence of phytol on liver lipids]. |
| PubMed: | Absorption and metabolism of uniformly 14C-labeled phytol and phytanic acid by the intestine of the rat studied with thoracic duct cannulation. |
| PubMed: | Gas chromatographic determination of phytol. |
| PubMed: | The incorporation of the two carbons of acetate and glycine into the phorbide and phytol moieties of chlorophylls a and b. |
| PubMed: | Metabolism of phytol-U-14C and phytanic acid-U-14C in the rat. |
| PubMed: | Effects of dietary phytol and phytanic acid in animals. |
| PubMed: | Phytol metabolism in the bovine. |
| PubMed: | The stereochemistry of phytol biosynthesis in the leaves of Ficus elasticus and Aesculus hippocastanum. |
| PubMed: | Conversion of H3-phytol to phytanic acid and its incorporation into plasma lipid fractions in heredopathia atactica polyneuritiformis. |
| PubMed: | Conversion of phytol into dihydrophytol and phytanic acid. |
| PubMed: | [Studies on the phytol metabolism in lipoidosis heredopathia atactica polyneuritiformis (Refsum's syndrome). The transformation of 3H-phytol into the 3,7,11,15-tetramethylhexadecanoic acid (phytanic acid) of the plasma lipoid fractions]. |
| PubMed: | Conversion of U-C14-phytol to phytanic acid and its oxidation in heredopathia atactica polyneuritiformis. |
| PubMed: | PHYTANIC ACID FORMATION AND ACCUMULATION IN PHYTOL-FED RATS. |
| PubMed: | [Studies on the metabolism of phytol, dihydrophytol and phytanic acid]. |
| PubMed: | RADIOCHEMICAL INVESTIGATION ON THE EXCHANGE BETWEEN PHYTOL AND PHYTYL OF CHLOROPHYLLS "A" AND "B" IN CHLORELLA VULGARIS. |
| PubMed: | INFLUENCE OF DIETARY PHYTOL, ISOPHYTOL, AND SQUALENE ON THE TOCOPHEROL CONTENT OF LIVER TISSUE. |
| PubMed: | [ISOLATION OF PHYTOL FROM PLANT MATERIAL AND ITS ANALYTICAL EVALUATION]. |
| PubMed: | [Phytol biosynthesis in the dark by green barley plants]. |
| PubMed: | [Role of glucose in formation of forbin and phytol part of chlorophyll molecule]. |
| PubMed: | [Pathology of fat metabolism; phytol, a new lipotropic factor of food]. |
| PubMed: | Effects of different amounts of lutein, squalene, phytol and related substances on the utilization of carotene and vitamin A for storage and growth in the rat. |
| PubMed: | THE CHEMICAL NATURE OF THE FAT-SOLUBLE VITAMIN OF GROWTH-PHYTOL, CAROTIN, VITAMIN A. |
|
 3D/inchi
3D/inchi





